 Thanks Again to and all for your help! I'm better now as a result. So now my calculation looks like this, where I have added the extra steps just to make it clear I'm playing fair with my units: (It also helps that it looks like these calculations were performed in Mathcad.) This was for a huge system in a fast river, so the values are quite high, but they handled specific weight and density correctly: Reading both of your posts made me question everything, so then I dug deeper into previous calculations done for our machines, and found this calculation, done by a structural engineering consultant. This was for a larger machine in a slightly faster flow, but I thought my values had to be in this ball park. The density of common metals such as iron is 7.87 g/cm3, mild steel is 7.85 g/cm3, 304 stainless steel is 8.0 g/cm3, aluminum is 2.7g/cm3, copper is 8.93 g/cm3, gold is 19.3 g/cm3, silver is 10.49 g/cm3, for more metals, please view the metal density chart and table below. You are exactly correct, in that I was looking at a hand calc example from my predecessor, and I was taking it as gospel, when in fact he was confusing specific weight and density. I hope this Thank You very much for your input, and sorry for the delay - I'm just super busy, trying to get this report completed for a project submittal. 
Yes - I wish the US would just drop this terrible unit system once and for all! The correct drag force should be about 30 lbf. Recall the numeric value of 1 pound of mass weighs (has a force of) 1 pound and is only valid in earth's gravity. The mass density of water is 62.4 pounds mass/ft^2. If you simply change the gamma unit tag in example #2 from "lbf" to "lb", you will get the correct result. Update - the equation you used for the drag force requires a mass density, not weight density.the Mathcad Prime result supports this. Densities using the following metric units all have exactly the same numerical value, one thousandth of the value in (kg/m 3 ). I've used this approach many times with great success. 
Rumor has it Prime 7 will have some controls.maybe a simple drop-down like we have in v15. You will need to standardize on a "density" unit and account for gravity, or not, in your equation.Īlternatively, if you use Mathcad 15, you could create a drop-down unit choice and use an "If" statement to set the equation up to handle the units based on user selection. Blanke 1 is used to calculate the density of water. thats 4 ounces more, lucky Brits.As you know, a "slug" is a different unit from a "lbf", so you can't use the same equation for both and expect consistent units for a result. s) Compute the product of the density of water, the velocity of the flow, and L: × u × L 249. 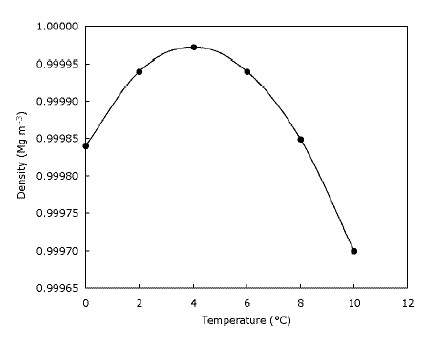
Find the dynamic viscosity: µ 0.001308 kg/(m To find this result: Find the density of water at 10 ☌: 999.7 kg/m³. Density of air at sea level / Density of water at 0☌ / Density of water at 4☌ / Density of water at 100☌ / Density of ice / Density of diamond. US Customary History of measurement Conversion questions. The source I used did not give temperature so even this is an estimate.īy the way, in the United States we do not use imperial gallons or imperial anything, at lest not since the revolution, we use a scale called avoirdupois I guess the originator of the thread was British? In avoirdupois the measurement is so close as to be meaningless in the home, not so in the Imperial system.īy the way, a pint of beer in the UK measures around 20 Fluid Ounces in the US. The Reynolds number for a water flow at u 1 m/s in an L 0.25 m pipe is: 191,074. Density Converter / Metric System / Milligram Per Cubic Meter mg/m³ Online converter page for a specific unit. Since Ounces weight (avoirdupois) and Fluid Ounces are actually totally unrelated scales (the term ounce has many meanings) there happens to be a nice accident so that a fluid ounce of water actually weighs 1.0425 ounces avoirdupois. The British gallon is defined as the volume of 10 Ibs of water of density 988.859 kg/ m 3 weighed in air with a density of 1. 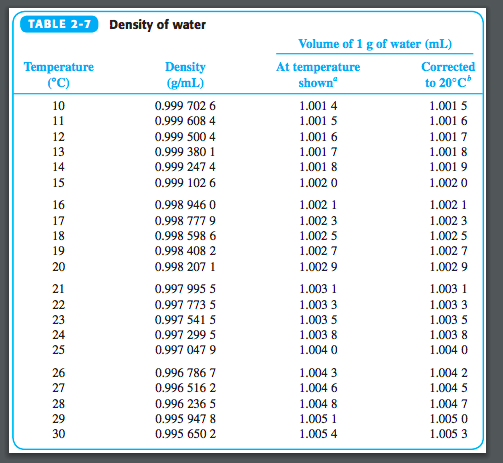
How technical and accurate do we need to be in baking? Here are the exact densities and, yes, temperature does matter, in analytical chemistry, not in baking.ġ milliliter (ml) = 1 cubic centimeter (cc) of pure water at 4 degrees centigrade - water's densist state (it expands on the phase change of freezing into a solid), so, 1 cubic centimeter contains 1 gram of water at 4 deg. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
